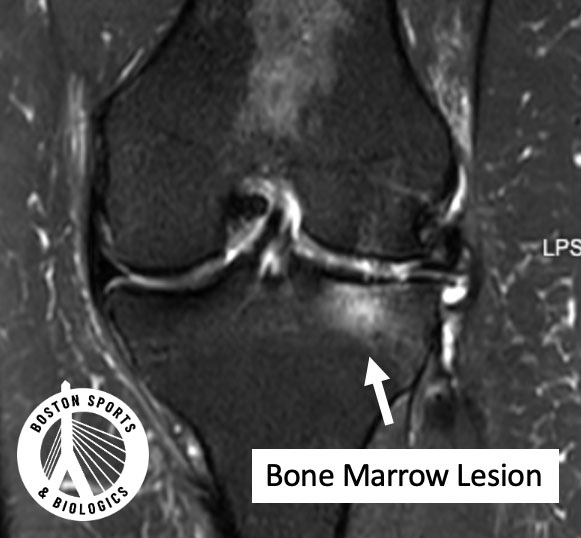
If you’ve been told you have a bone marrow lesion (BML) on your MRI, you’re not alone. These lesions are common in knee osteoarthritis (OA) and are strongly associated with pain, faster cartilage loss, and progression toward joint replacement. Learn more about bone marrow lesions here.
At Boston Sports & Biologics, we frequently evaluate patients whose MRI shows subchondral bone changes driving their symptoms. Increasingly, we’re using bone marrow aspirate concentrate (BMAC) injections delivered directly into the subchondral bone to address this pathology at its source.
Let’s break down the mechanism behind how BMAC may work for bone marrow lesions. Learn more about BMAC here.
Bone marrow lesions (sometimes called “bone marrow edema”) appear on MRI as areas of increased signal in the subchondral bone.
Histologically, they are not simply fluid. They often represent:
Microfractures of trabecular bone
Increased vascularity and capillary leakage
Fibrovascular tissue replacement of marrow
Elevated bone turnover
Nerve ingrowth
Bone Marrow death (nerosis)
These changes correlate strongly with pain and disease progression. In fact, patients with BMLs are significantly more likely to progress to knee replacement than those without them (Tanamas et al, 2010; Nielsen et al, 2017; Raynauld et al, 2011).
Importantly, this means:
Osteoarthritis is not just a cartilage disease.
It is a disease of the entire joint—including the bone beneath the cartilage.
Traditional treatments focus on decreasing inflammation with corticosteroid injections,
Hyaluronic acid injection or intra-articular platelet rich plasma (PRP) injections.
These can help symptoms—but they do not directly address subchondral bone pathology. Emerging research suggests that in some patients, OA may begin in the bone and progress upward toward cartilage (Funck-Brentano & Cohen-Solal, 2015; Hügle & Geurts, 2017).
When bone remodeling becomes dysregulated, it leads to:
Abnormal angiogenesis
Increased osteoclast activity
Structural instability
Pain mediator release
That’s where intraosseous (IO) biologic injections come in.
Bone marrow aspirate concentrate (BMAC) is derived from your own bone marrow (typically from the iliac crest).
After processing, it contains:
Hematopoietic progenitors
Platelets
Growth factors
Cytokines
Angiogenic mediators
Studies show that concentration systems can increase MSC yield approximately 4-fold compared to raw aspirate, while preserving cell viability and differentiation potential.
In a 2024 rat osteoarthritis model (Zhang et al., AJSM), combined intra-articular + intraosseous biologic injections:
Reduced abnormal CD31hiEmcnhi vessel formation
Reduced excessive osteoclast and osteoblast turnover
Preserved subchondral microarchitecture
Slowed cartilage degeneration
This suggests that subchondral biologics may:
✔ Normalize pathologic bone remodeling
✔ Stabilize trabecular bone
✔ Interrupt the vicious cycle of bone-cartilage degeneration
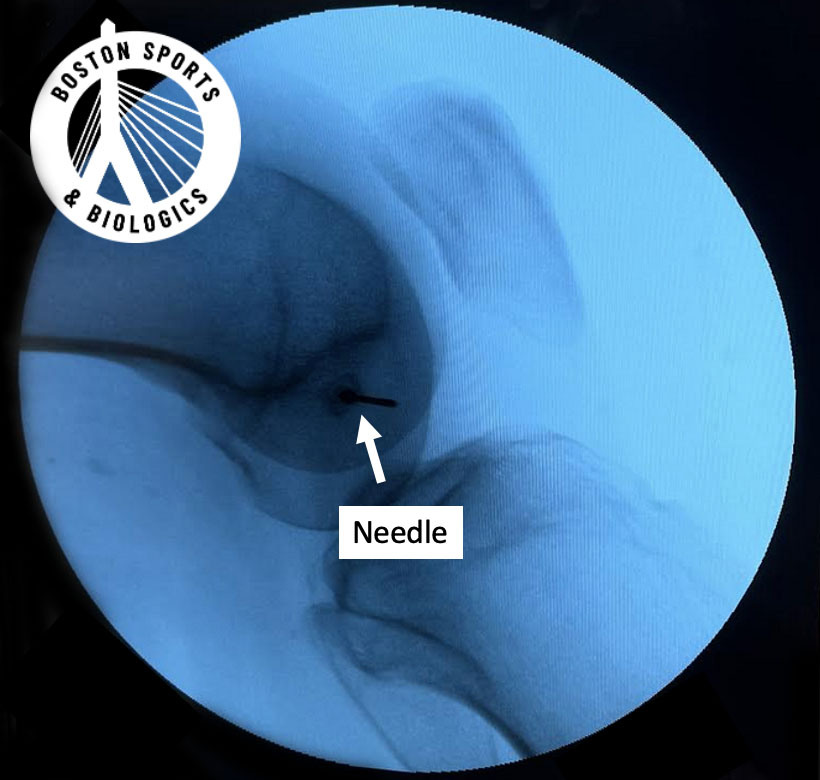
Subchondral bone is richly innervated.
The same study by Zhang et al., AJSM, they demonstrated that IO biologic injections reduced:
Calcitonin gene-related peptide (CGRP)
Substance P
These are neuropeptides strongly associated with pain signaling.
This supports the idea that:
BML-associated pain is partly driven by subchondral nociceptive activity.
By modulating this environment, BMAC may reduce pain at a deeper structural level—not just by suppressing inflammation.
MSCs in BMAC act less like “replacement cells” and more like cell factories. They secrete VEGF, BMP-2, IL-6, IL-8, angiopoietin-1, and TGF-β.
These mediators can:
Modulate inflammation
Reduce apoptosis
Support vascular normalization
Promote tissue repair
Importantly, MSCs appear to help restore a healthier marrow microenvironment, rather than simply adding new cells (Fu et al, 2017; Baberg et al, 2019).
Human data has shown that intraosseous PRP can alter the number of mesenchymal stem cells present in synovial fluid—suggesting that the subchondral bone and joint space communicate dynamically.
In osteoarthritis, there is abnormal signaling between:
Subchondral bone
Cartilage
Synovium
Targeting subchondral bone not only affects the joint space but also remodels the bone marrow microenvironment itself (Ganguly et al, 2022).
Targeting bone resets pathologic crosstalk. The therapeutic rationale is that normalizing subchondral bone microenvironment can interrupt the vicious cycle of bone-cartilage-synovium degeneration (Zhu et al, 2021; Gardner et al, 2022; Hu et al, 2021).
Bone marrow lesions are not incidental findings—they are active drivers of osteoarthritis progression and pain.
BMAC injections delivered into the subchondral bone may work by:
Normalizing abnormal bone remodeling
Suppressing pain mediators
Modulating inflammatory signaling
Restoring marrow microenvironment health
Improving bone-cartilage cross-talk
As our understanding of OA evolves, so must our treatment strategies.
At Boston Sports & Biologics, we focus on precision orthobiologic approaches—targeting the structures actually driving symptoms.
If you’ve been told you have a bone marrow lesion and want to explore options beyond surgery, we’re happy to review your imaging and discuss whether subchondral biologic therapy may be appropriate.
Baberg F, Geyh S, Waldera-Lupa D, Stefanski A, Zilkens C, Haas R, Schroeder T, Stühler K. Secretome analysis of human bone marrow derived mesenchymal stromal cells. Biochim Biophys Acta Proteins Proteom. 2019 Apr;1867(4):434-441. doi: 10.1016/j.bbapap.2019.01.013. Epub 2019 Feb 1. PMID: 30716505.
Fu Y, Karbaat L, Wu L, Leijten J, Both SK, Karperien M. Trophic Effects of Mesenchymal Stem Cells in Tissue Regeneration. Tissue Eng Part B Rev. 2017 Dec;23(6):515-528. doi: 10.1089/ten.TEB.2016.0365. PMID: 28490258.
Funck-Brentano T, Cohen-Solal M. Subchondral bone and osteoarthritis. Curr Opin Rheumatol. 2015 Jul;27(4):420-6. doi:10.1097/BOR.0000000000000181. PMID: 26002035.
Ganguly P, Fiz N, Beitia M, Owston HE, Delgado D, Jones E, Sánchez M. Effect of Combined Intraosseous and Intraarticular Infiltrations of Autologous Platelet-Rich Plasma on Subchondral Bone Marrow Mesenchymal Stromal Cells from Patients with Hip Osteoarthritis. J Clin Med. 2022 Jul 4;11(13):3891. doi: 10.3390/jcm11133891. PMID: 35807175; PMCID: PMC9267269.
Gardner JE, Williams CW, Bowers RL. Subchondral versus intra-articular orthobiologic injections for the treatment of knee osteoarthritis: a review. Regen Med. 2022 Jun;17(6):389-400. doi: 10.2217/rme-2021-0174. Epub 2022 Apr 12. PMID: 35410486.
Hu W, Chen Y, Dou C, Dong S. Microenvironment in subchondral bone: predominant regulator for the treatment of osteoarthritis. Ann Rheum Dis. 2021 Apr;80(4):413-422. doi: 10.1136/annrheumdis-2020-218089. Epub 2020 Nov 6. PMID: 33158879; PMCID:PMC7958096.
Hügle T, Geurts J. What drives osteoarthritis?-synovial versus subchondral bone pathology. Rheumatology (Oxford). 2017 Sep 1;56(9):1461-1471. doi: 10.1093/rheumatology/kew389. PMID: 28003493.
Nielsen FK, Egund N, Jørgensen A, Jurik AG. Risk factors for joint replacement in knee osteoarthritis; a 15-year follow-up study. BMC Musculoskelet Disord. 2017 Dec 4;18(1):510. doi: 10.1186/s12891-017-1871-z. PMID: 29202806; PMCID: PMC5715644.
Raynauld JP, Martel-Pelletier J, Haraoui B, Choquette D, Dorais M, Wildi LM, Abram F, Pelletier JP; Canadian Licofelone Study Group. Risk factors predictive of joint replacement in a 2-year multicentre clinical trial in knee osteoarthritis using MRI: results from over 6 years of observation. Ann Rheum Dis. 2011 Aug;70(8):1382-8. doi: 10.1136/ard.2010.146407. Epub 2011 May 8. PMID: 21551506.
Schäfer R, DeBaun MR, Fleck E, Centeno CJ, Kraft D, Leibacher J, Bieback K, Seifried E, Dragoo JL. Quantitation of progenitor cell populations and growth factors after bone marrow aspirate concentration. J Transl Med. 2019 Apr 8;17(1):115. doi: 10.1186/s12967-019-1866-7. PMID: 30961655; PMCID: PMC6454687.
Tanamas SK, Wluka AE, Pelletier JP, Pelletier JM, Abram F, Berry PA, Wang Y, Jones G, Cicuttini FM. Bone marrow lesions in people with knee osteoarthritis predict progression of disease and joint replacement: a longitudinal study. Rheumatology (Oxford). 2010 Dec;49(12):2413-9. doi: 10.1093/rheumatology/keq286. Epub2010 Sep 7. PMID: 20823092.
Zhang K, Yu J, Li J, Fu W. The Combined Intraosseous Administration of Orthobiologics Outperformed Isolated Intra-articular Injections in Alleviating Pain and Cartilage Degeneration in a Rat Model of MIA-Induced Knee Osteoarthritis. Am J Sports Med. 2024Jan; 52(1):140-154. doi: 10.1177/03635465231212668. PMID: 38164685.
Zhu X, Chan YT, Yung PSH, Tuan RS, Jiang Y. Subchondral Bone Remodeling: A Therapeutic Target for Osteoarthritis. Front Cell Dev Biol. 2021 Jan 21;8:607764. doi: 10.3389/fcell.2020.607764. PMID: 33553146; PMCID: PMC7859330.
Ultrasound-guided tendon scraping is an emerging minimally invasive treatment for patellar tendinitis (jumper’s knee). A recent study shows promising results with faster return to sport and significant pain relief,
Read MorePatellar tendinitis (“jumper’s knee”) is a common cause of knee pain in athletes. Learn what causes it, why it lingers, and how advanced treatments like ultrasound-guided tendon scraping may help you recover faster and
Read More